- 24 healthy volunteers have been dosed in study SB102 to assess the safety, PK, and PD of SON-1010 without the background of prior chemotherapy
- PK data shows an extended half-life of SON-1010 of approximately 112 hours, compared to 12 hours for rhIL-12
- Simulation of the controlled and prolonged induction of IFNg and minimal related adverse events suggest that dose escalation can continue
PRINCETON, NJ / ACCESSWIRE / January 19, 2023 / Sonnet BioTherapeutics Holdings, Inc. (NASDAQ:SONN) (the "Company" or "Sonnet"), a clinical-stage company developing targeted immunotherapeutic drugs, announced today that pharmacokinetic (PK) profile simulation of SON-1010 dosing has been completed in its randomized, placebo-controlled Phase 1 clinical trial in healthy volunteers. Historically, the therapeutic application of cytokines has been limited by relatively short half-lives and off-target toxicities that are typically associated with peak plasma levels. SON-1010 is a proprietary version of recombinant human interleukin-12 (rhIL‑12), configured using Sonnet's Fully Human Albumin Binding (FHAB®) technology, which has been shown to extend PK and to reduce peak drug levels for improving pharmacodynamic (PD) toxicity. The FHAB technology was designed to promote targeting to the tumor microenvironment (TME), particularly when levels of Secreted Protein Acidic and Rich in Cysteine (SPARC) are elevated in the TME. Study SB102 is a single-ascending dose trial in healthy volunteers (NCT05408572) that was initiated in July, 2022 to address the safety, PK, and PD of SON-1010 in subjects without interference from prior chemotherapy. The Safety Review Committee has found no significant safety concerns to date and has approved advancing to each higher dose level.
"We have evaluated the drug levels and cytokine responses in the SB102 study," said Richard Kenney, M.D., Sonnet's Chief Medical Officer. "The PK and PD from 3 cohorts have been formally simulated to predict the levels of the most responsive cytokines. Interferon gamma (IFNg) levels rose after SON-1010 dosing as expected, but with lower peak elevations and much longer decay than those observed historically for rhIL-12, which correlates with better tumor control in preclinical models. Linking IL-12 to the FHAB is designed to permit use of higher doses of this cytokine without triggering unacceptable toxicity, allowing us to safely induce a successful local immune response to IL-12 in the TME."
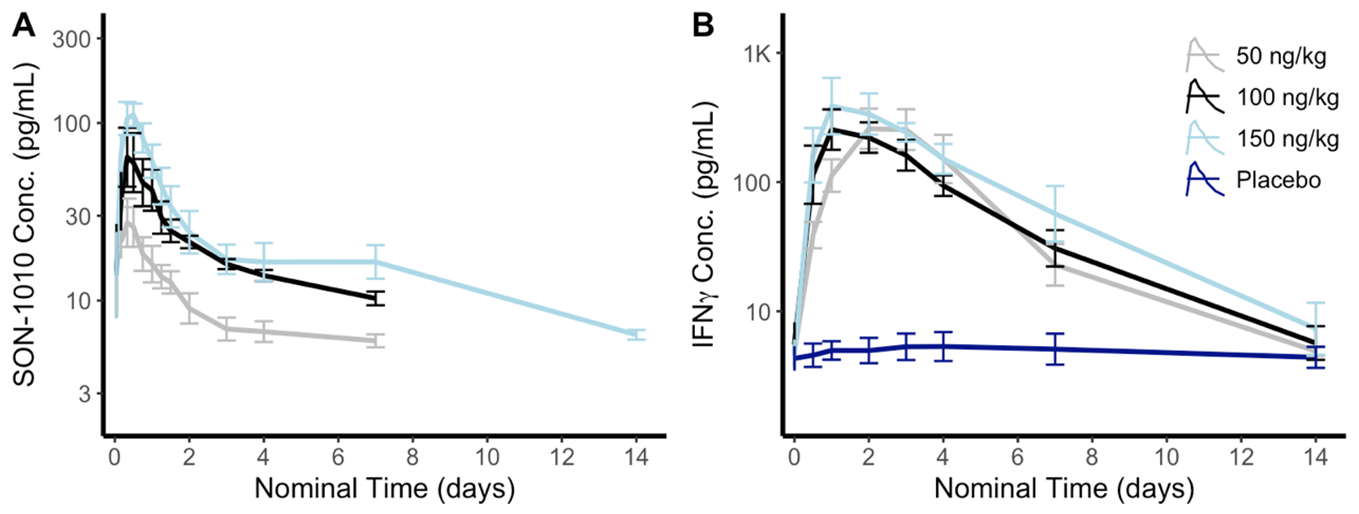
Typical dose-related increases were seen with SON-1010 using a validated electrochemiluminescence assay (Meso Scale Discovery®) after subcutaneous administration. Drug levels peaked at about 11 hours with a geometric mean maximum concentration (Cmax) of 29, 68, and 125 pg/mL for the 50, 100, and 150 ng/kg dose groups, respectively (Figure 1). The mean elimination half-life (T½) after a 150 ng/kg dose of SON-1010 was 112 hours, compared to 12 hours for rhIL-12.
Observed increases in IFNg were most pronounced and were dose-related, controlled, and prolonged. SON-1010 induced IFNg in all active-drug subjects, which peaked at 24 to 48 hours then returned to baseline after 2 weeks (Figure 2). The IFNg geomean Cmax was 398, 384, and 666 pg/mL after 50, 100, or 150 ng/kg of SON-1010, respectively, while the AUC over 48 hours was 6050, 10200, and 14600 h*pg/mL. Linear regression was used to predict the IFNg Cmax at higher doses, which remain well within the range of safety. Low amounts of IL-10 were induced in a dose-dependent manner, which could also be due to the increase in IFNg. There were small transient increases in IL-6, IL‑8, and TNFα after dosing but no consistent pattern was seen with IL-1β, IL-2, or IL-4 and there was no evidence of cytokine release syndrome (CRS). Safety was consistent with what has been reported previously; adverse events have generally been mild/moderate, transient in nature, and have all been tolerable.
Figure 1 and Figure 2
"This demonstration of extended PK with SON-1010 in humans, in conjunction with the prior preclinical tumor accumulation data, represents a significant step forward in Sonnet's approach to immunotherapy with the FHAB platform. We believe, based on the totality of the data we have generated to this point, that we have a best-in-class IL-12 molecule with synergistic potential in cancer and clear business development opportunities to explore." said Pankaj Mohan, Ph.D., Sonnet Founder and Chief Executive Officer. "The half-life of SON-1010 fits well with 3-week checkpoint inhibitor dosing schedules, which supports the plans for the SB221 combination study that we announced earlier this month."
About SON-1010
SON-1010 is a candidate immunotherapeutic recombinant drug that links unmodified single-chain human IL-12 with the albumin-binding domain of the single-chain antibody fragment A10m3. This was selected to bind both at normal pH, as well as at an acidic pH typically found in the TME. The FHAB technology targets tumor and lymphatic tissue, providing a mechanism for dose sparing and an opportunity to improve the safety and efficacy profile of not only IL-12, but a variety of potent immunomodulators. Interleukin-12 can orchestrate a robust immune response to many cancers and pathogens. Given the types of proteins induced in the TME, such as the Secreted Protein and Rich in Cysteine (SPARC) and glycoprotein 60 (GP60), several types of cancer such as non-small cell lung cancer, melanoma, head and neck cancer, sarcoma, and some gynecological cancers are particularly relevant for this approach. SON-1010 is designed to deliver IL-12 to local tumor tissue, turning ‘cold' tumors ‘hot' by stimulating IFNg, which activates innate and adaptive immune cells and increases the production of Programed Death Ligand 1 (PD-L1) on tumor cells.
About the SB102 Phase 1 Trial
The SB102 study is designed to robustly evaluate the safety, PK and PD of single ascending doses of SON-1010, using larger groups of healthy volunteers, and is being conducted at a single site in Australia. The study is done in a blinded fashion, comparing a single dose of SON-1010 to placebo utilizing up to five cohorts. Both PK and PD will be closely followed during dose escalation in this double-blind, placebo-controlled study, along with an assessment of the cellular immune responses at each dose using sophisticated fluorescence activated cell sorting (FACS) analysis. The primary endpoint explores the safety and tolerability of SON-1010, with key secondary endpoints intended to measure PK, PD, and immunogenicity.
About Sonnet BioTherapeutics Holdings, Inc.
Sonnet BioTherapeutics is an oncology-focused biotechnology company with a proprietary platform for innovating biologic drugs of single or bispecific, bifunctional action. Known as FHAB (Fully Human Albumin Binding), the technology utilizes a fully human single chain antibody fragment (scFv) that binds to and "hitch-hikes" on human serum albumin (HSA) for transport to target tissues. Sonnet's FHAB was designed to specifically target tumor and lymphatic tissue, with an improved therapeutic window for optimizing the safety and efficacy of immune modulating biologic drugs. FHAB is the foundation of a modular, plug-and-play construct for potentiating a range of large molecule therapeutic classes, including cytokines, peptides, antibodies, and vaccines.
Forward-Looking Statements
This press release contains certain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934 and Private Securities Litigation Reform Act, as amended, including those relating to the Company's product development, clinical and regulatory timelines, market opportunity, competitive position, possible or assumed future results of operations, business strategies, potential growth opportunities and other statements that are predictive in nature. These forward-looking statements are based on current expectations, estimates, forecasts and projections about the industry and markets in which we operate and management's current beliefs and assumptions.
These statements may be identified by the use of forward-looking expressions, including, but not limited to, "expect," "anticipate," "intend," "plan," "believe," "estimate," "potential, "predict," "project," "should," "would" and similar expressions and the negatives of those terms. These statements relate to future events or our financial performance and involve known and unknown risks, uncertainties, and other factors which may cause actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. Such factors include those set forth in the Company's filings with the Securities and Exchange Commission. Prospective investors are cautioned not to place undue reliance on such forward-looking statements, which speak only as of the date of this press release. The Company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise.
Sonnet BioTherapeutics Investor Contact:
Michael V. Morabito, Ph.D.
Solebury Strategic Communications
917-936-8430
[email protected]
SOURCE: Sonnet BioTherapeutics, Inc.




