Game changing technology actively neutralizes surface and airborne pathogens including SARS-CoV-2
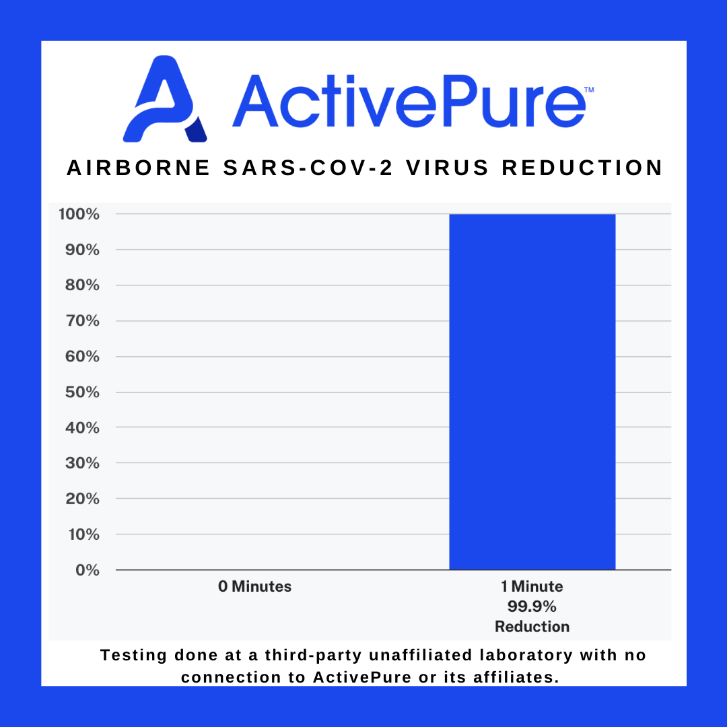
DALLAS, TX / ACCESSWIRE / March 8, 2022 / New independent testing conducted by the University of Texas Medical Branch (UTMB), one of the world's top biosafety testing facilities, shows that ActivePure Technology inactivated over 99.9% of highly concentrated airborne SARS-CoV-2 virus-the virus that causes COVID-19-in an enclosed setting to below detectable levels in a 60 second testing period. This recent testing round shows definitively that the technology works even faster than previously indicated. ActivePure's purifying technology provides 24/7 full-coverage air and surface disinfection by deactivating pathogens in the air and on surfaces.
In 2020 after the first UTMB test, Dr. William S. Lawrence, Ph.D., director of the Aerobiology Service Center at UTMB's Galveston National Laboratory, said, "These results clearly demonstrate the effectiveness of this technology in a laboratory setting." With ActivePure's latest test results, he said, "Importantly, the device is still showing a very high level of inactivation after just one minute."
In the wake of the pandemic, ActivePure has become the air purification solution of choice worldwide for commercial, residential, educational, and medical settings where pure air and surfaces are required. While ActivePure has been on the worldwide retail market as an air purifier for over ten years, its commercial use has become more prevalent during the pandemic.
"In addition to our independent testing data, we have real-world data that specifically looks at how our customer's outcomes improved based on ActivePure, which further demonstrates our technology's efficacy," said Joe Urso, chairman and CEO of ActivePure Technologies, LLC. "When it comes to SARS-CoV-2, we know that multi-layered protection strategies work the best to mitigate disease and protect occupants. ActivePure has become an important part of this multi-layered approach."
The tests were done in triplicate. To determine the effectiveness of ActivePure, the live SARS-CoV-2 virus was sprayed into a test chamber at extremely high concentrations (7+ logs, or over 10 million particles per milliliter). In less than 60 seconds, the air purification and disinfection calibrated to the lowest setting of only 29 cubic feet per minute of air movement inactivated the concentration of the virus to below detectable limits of 1.6 logs, a reduction of 99.57 to >= 99.83%% but overall net reduction likely exceeded 99.99%. The testing products only used ActivePure; all other purification technologies found within the testing product were disabled or removed.
Developed over the past 27 years, ActivePure has successfully adapted what happens naturally outdoors for indoor environments. ActivePure is an active purification technology that uses Advanced Photocatalysis. Advanced Photocatalysis technology mimics the organic process of photolysis - in this process, the air is cleansed when sunlight interacts with moisture and creates oxidative molecules. ActivePure brings this atmospheric cleansing phenomenon indoors, allowing the oxidizing particles to neutralize viruses, mold and bacteria in the air and on surfaces. This unique technology is a game-changer as it actively neutralizes surface and airborne pathogens (including SARS-CoV-2) without chemicals, ozone and without the need to trap or filter contaminants.
ActivePure is affordable and customizable. More than 75 products are powered by ActivePure, from standalone portable products to products installed within heating and air conditioning units to handheld travel products. In addition, ActivePure units run 24/7 and can easily be combined for a custom solution serving specific spaces' unique needs.
To learn more about ActivePure, its uses and scientific studies on its proven effectiveness, please visit ActivePure.comor ActivePureMedical.com or call 888-217-4316.
###
ABOUT ACTIVEPURE
Privately held ActivePure has been the global leader in active, continuous surface and air disinfection systems for health care and educational institutions, commercial and public facilities, hospitality and residential applications since 1924. Patented ActivePure Technology has been proven in independent university and laboratory testing to effectively control and neutralize indoor contaminants. It is the only product in its class recognized by the Space Foundation as Certified Space Technology and inducted into the Space Foundation Hall of Fame. In addition, the ActivePure Medical Guardian is registered and cleared as an FDA Class II Medical Device. ActivePure was developed for use in space exploration and has since evolved for use in commercial and consumer products used to reduce exposure to pathogens, including RNA and DNA viruses, bacteria and molds, by up to 99.9% in the air and on surfaces. For more information, please visit ActivePure.com or call 888-217-4316.
MEDIA CONTACTS:
Jo Trizila, TrizCom PR on behalf of ActivePure
Email: [email protected]
Office: 972-247-1369
Cell/Text: 214-232-0078
Tonie Auer, TrizCom PR on behalf of ActivePure
Email: [email protected]
Office: 972-247-1369
Cell/Text: 817-925-2013
SOURCE: ActivePure Technology




